Medical-grade, hypoallergenic, and skin-safe adhesive tapes are widely used in healthcare and wearable devices—but what do these terms actually mean?
While these labels are not strictly regulated, they typically indicate that the adhesive material has undergone biocompatibility testing to ensure it is safe for contact with human skin. This includes evaluating the risk of skin irritation, cytotoxicity, and sensitization, often following internationally recognized standards such as ISO 10993.
What Is Medical-Grade Adhesive Tape?
Medical-grade adhesive tape refers to adhesive materials designed for use on or around the human body. These tapes are engineered to meet biocompatibility requirements, making them suitable for applications such as:
- Wearable medical devices
- Wound care products
- Skin-contact sensors
- Long-term skin adhesion
Although the term “medical-grade” is not tightly regulated, it is commonly supported by standardized laboratory testing.
What Does Hypoallergenic and Skin-Safe Tape Mean?
- Hypoallergenic adhesive tape is designed to minimize the risk of allergic reactions.
- Skin-safe adhesive indicates reduced potential for irritation during skin contact.
Both claims are typically backed by biocompatibility testing, rather than formal regulatory
Biocompatibility Testing for Adhesive Tapes
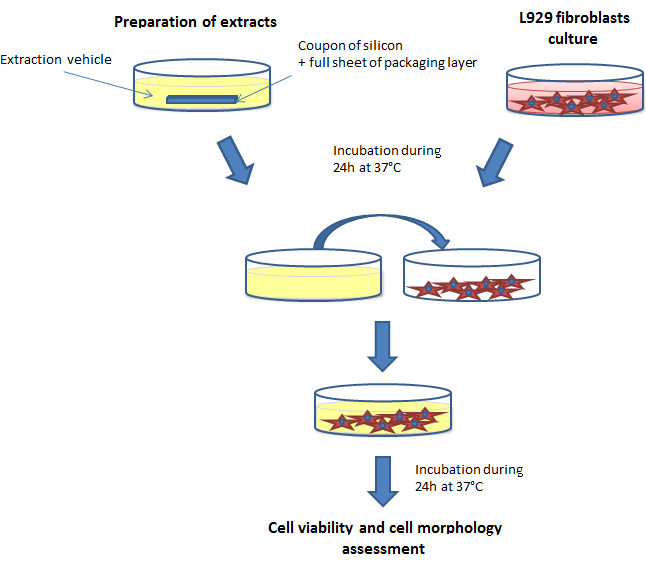
Biocompatibility testing evaluates whether a material is safe for human use. For adhesive tapes, this includes assessing how the material interacts with skin and cells.
Cytotoxicity Testing (ISO 10993-5)
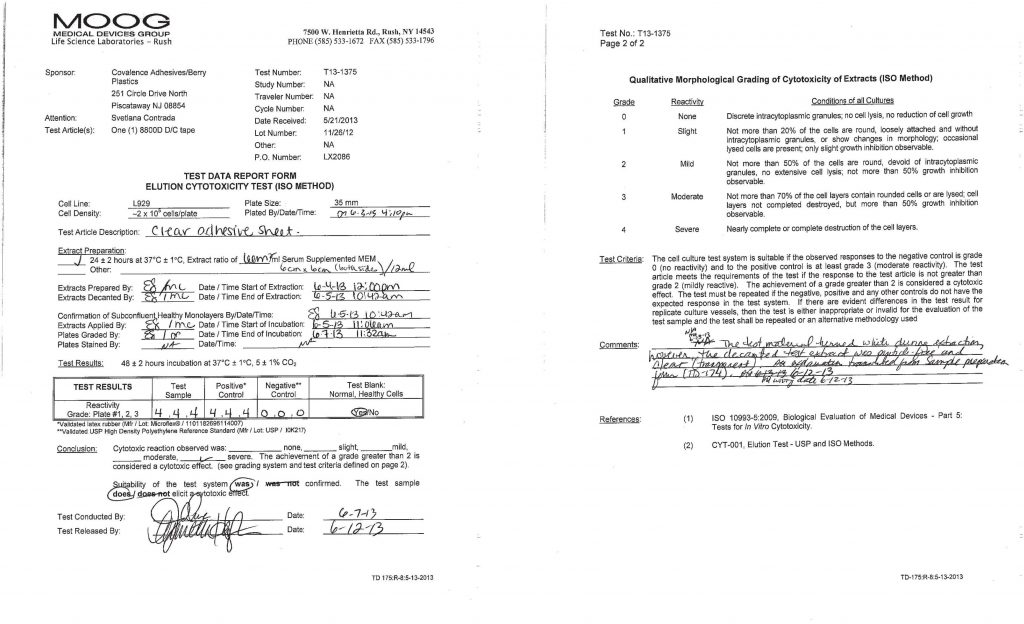
Cytotoxicity testing determines whether an adhesive releases substances that are toxic to cells.
How it works:
- Adhesive samples are prepared
- Materials are extracted using a solvent
- Cultured cells (such as fibroblasts or epithelial cells) are exposed to the extract
- Cells are evaluated after exposure (typically 48 hours)
- Cell viability and morphology are analyzed
- Results are graded based on cytotoxic response
This method is conducted in a laboratory and does not involve human subjects.
Cytotoxicity Grading Scale (ISO 10993-5)
Materials are graded on a scale from 0 to 4:
- Grade 0: No cytotoxic response
- Grade 1: Slight reactivity
- Grade 2: Mild reactivity
- Grade 3: Moderate reactivity
- Grade 4: Severe reactivity
Grades 0–2 are generally considered acceptable for medical applications.
Human Repeat Insult Patch Test (RIPT)
The Human Repeat Insult Patch Test (RIPT) evaluates skin irritation and sensitization in human volunteers.
- Conducted over 4–6 weeks
- Involves repeated application of the adhesive
- Measures cumulative skin response
RIPT is commonly used for finished medical devices and consumer skin-contact products.
Industry Practices: Adhesive vs. Medical Device Testing
Testing requirements vary depending on the application:
- Adhesive tape manufacturers typically rely on cytotoxicity testing (ISO 10993-5) due to its efficiency and standardized approach.
- Medical device manufacturers often include RIPT testing when evaluating complete devices for regulatory approval.
For double-sided adhesive tapes, each adhesive surface may be tested independently—especially when different chemistries (such as acrylic and silicone adhesives) are used.
Example of testing showing adhesive tape not viable for medical skin application
Why Biocompatibility Matters for Medical Adhesives
Biocompatibility testing helps ensure that adhesive tapes:
- Are safe for prolonged skin contact
- Minimize irritation and allergic reactions
- Meet industry expectations for medical use
- Support regulatory submissions for medical devices
This is especially critical for wearable and long-term applications, where adhesives remain in contact with skin for extended periods.
Our Approach to Medical-Grade Adhesive Tape
We work with independent laboratories to conduct cytotoxicity testing using ISO-standard methods. Our double-sided adhesive constructions are engineered to meet widely accepted biocompatibility criteria, making them suitable for medical and skin-contact applications.
Key Takeaways
- “Medical-grade” and “hypoallergenic” typically indicate biocompatibility testing, not strict regulation
- ISO 10993-5 cytotoxicity testing is the most common evaluation method
- RIPT testing is used for human skin response assessment
- Grades 0–2 in cytotoxicity testing are considered safe for medical use
- Double-sided adhesives may require independent testing of each surface